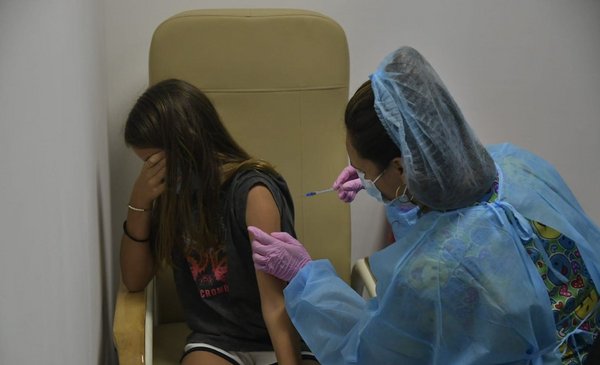
This Wednesday, January 12, the vaccination of children between 5 and 11 years old began and in this way Uruguay joins the list of countries that decided to inoculate children.
The members of this age range who will be vaccinated with two pediatric doses from the Pfizer laboratory and, so far, a third of this population was scheduled to be vaccinated, approximately 116 thousand peopleAccording to the numbers that the president, Luis Lacalle Pou, and the Minister of Health, Daniel Salinas, shared at a press conference on Monday, the 10th.
12,700 minors are scheduled for this Wednesday, who should attend accompanied by an adult, As reported The country. The vaccine it is not mandatory, and in case of having passed through the coronavirus the minor you will have to wait three months to get vaccinated.
Children are the last strip to enter a vaccination campaign that until this Wednesday has a 78.40% of the population with at least one dose of the covid vaccine, 75.66% with two doses and 44.80% with a booster dose. These numbers, which place Uruguay among the countries with the highest coverage against covid-19, are reflected in a low occupancy of severe covid cases in intensive care centers and low number of deaths, in the middle of the largest wave of infections due to the arrival of omicron.
On January 11 they reported 49,373 active cases in the country, but there are 46 people in CTI and three new deaths were confirmed, numbers far from those seen in the wave of cases in the first half of 2021.
Despite the fact that children were never present in the statistics of serious cases – except for exceptions such as the Multisystemic Inflammatory Syndrome (MIS-C) that affects children who have had the virus – the need to vaccinate this group arose when, due to the high vaccination of the adult population, the vectors of contagion became the smallest. This led to an increase in cases in October due to outbreaks occurred in different schools, which at the time even stopped the government’s plans for complete the health emergency “as soon as possible”.
On November 24, the National Vaccination Advisory Commission (CNAV) approved the inoculation of minors between 5 and 11 years of age with Pfizer, and on December 29 the agenda was opened, the same day that the first 54 thousand doses for this group arrived in the country. The intention of the authorities is “arrive at March 7”, the date of the beginning of classes, “with the highest number of immunized gurises”, said the Secretary of the Presidency, Álvaro Delgado, in a press conference on December 28.
recommendations
EFE
Before the vaccine ASSE called to “do not administer analgesics” to minors, and to inform vaccinators if the child to be vaccinated has allergies
The State Health Services Administration (ASSE) published on its Twitter this Tuesday a series of recommendations for children who are vaccinated against the coronavirus. The agency recalled that If the minor received another vaccine more than 14 days ago, they can be vaccinated.
Before the ASSE vaccine called “do not administer painkillers” to minors, and to inform vaccinators if the child to be vaccinated has allergies. After the vaccination the child must remain in the vaccination center between 15 and 30 minutes, just like adults.
The health agency warned that adverse effects can be “pain, redness, swelling” at the vaccine site Y “tiredness, headache, muscle pain, chills, fever, nausea” in the body.
Advantages
AFP
In the United States, 15 cases of myocarditis were detected in 8 million vaccinated minors, explained the pediatrician Pujadas
“We have excellent evidence of efficacy and safety. We have a favorable benefit-risk analysis.”said Beth Bell, an infectious disease specialist and member of the expert committee convened by the Centers for Disease Control and Prevention (CDC), in an interview with AFP, when On November 2, this body authorized the vaccination with Pfizer in the United States. At that time, studies indicated that this vaccine prevents symptomatic disease in children by 90.7%.
Nowadays 61 countries vaccinate children against covid. The United States has nine million children vaccinated, of which less than 0.05% had adverse reactions, within which 97% were mild symptoms as a arm pain or a passing fever, as reported by the CDC.
The day after the approval of the vaccine for children in Uruguay, Minister Salinas valued in a tweet that pediatric dose “avoid absenteeism” in education, something that “impacts on educational quality (through connectivity)” and also in “the food they receive at school”.
Another not minor issue is that vaccination avoids absenteeism, or not being present, especially in vulnerable populations, impacts on educational quality (through connectivity) and eventually the food they receive at school.
– Daniel Salinas (@DrDanielSalinas) November 25, 2021
In an interview with the radio program In perspective (Radiomundo) on January 5, the pediatric epidemiologist Mónica Pujadas, a member of the CNAV, assured that the vaccination of this group “it is one more link in the immunization chain”, with a vaccine that has “millions of doses applied in the world”, and a “good safety profile”.
Pujadas assured that in the balance of risks and benefits “there are more benefits”. He explained that in the United States 15 cases of myocarditis in 8 million vaccinated minors, and everyone were recovered or on the mend. “The adverse effects have been inflammation and discomfort at the site of the puncture, but there are no serious adverse effects”, he concluded.
Pediatric dose
Pfizer’s pediatric coronavirus vaccine has three times less fluid than the dose given for adults. While the traditional vaccine has 30 µg (micrograms), the one for children has 10 µg.
Between the first and second doses, a waiting time of six weeks was marked, longer than expected for adults.. The recommendation was for a minimum of three weeks, and a maximum of a month and a half.