The National Institute of Hygiene indicated that if a patient identifies that they are using herceptin from counterfeit batches, they should immediately discontinue its use, due to the risks it may pose to health.
The National Institute of Hygiene Dr. Rafael Rangel issued a health alert after detecting the circulation of counterfeit batches of the drug herceptin (trastuzumab) in the country, used in the treatment of some types of cancer.
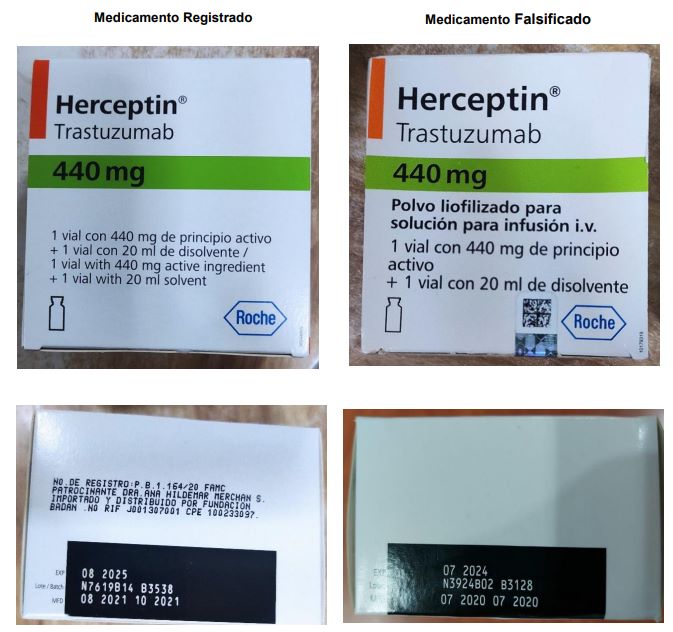
According to health alert No. 001/2023dated March 8, 2023, the investigations and analyzes carried out revealed that batches N3924B02 and N3927 of herceptin (trastuzumab) of 440 milligrams, in lyophilized powder for intravenous infusion PB 1,164/20, found in Venezuela are counterfeit products that represent a health risk.
“When carrying out the quality control tests on the questioned samples, it was determined that do not contain the active substance trastuzumabconcluding that the batches of the product are non-compliant for use and consumption, “explained the Institute in its alert document.
According to the agency, the company that owns the original product (Roche Laboratories), through its representative in Venezuela (Roche Products), stated that the batches identified with the numbers N3924B02 and N3927 were not manufactured by them. Additionally, lot N3927 differs from the original packaging art used by the lab.
Organizations and patients They have denounced constant failures in the delivery of trastuzumab by the Venezuelan Institute of Social Security (IVSS). In the private sector, a single vial can cost up to $1,850.
Herceptin is used to treat some types of cancer.
If any patient identifies that they are using any of the products described, the agency requested immediately discontinue usedue to the risks it can pose to health.
The Institute reported that the pharmacies of the BADAN Antineoplastic Drug Bank (Badan Foundation) they are the only ones that dispense the actual product.
«If it is acquired outside the indicated pharmacies, notify the INHRR through the website http://inhrr.gob.vey/or email alertasinhrr@gmail.com to validate its authenticity,” said the agency, which also urged those places or people who distribute or market counterfeit products to denounce through the same email.


















