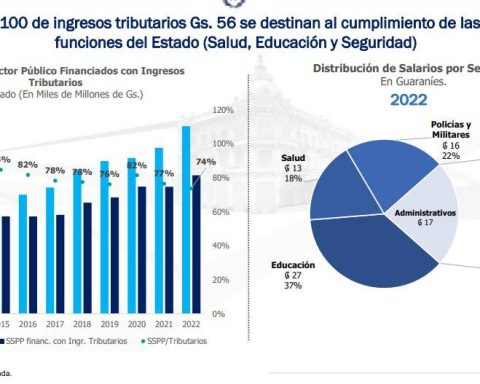
Here are the answers to the most frequently asked questions:
1. What are the documents to present for vaccination in children and adolescents?
The required documents are:
• Identity card of the adult and the child or adolescent.
• Photocopy of birth certificate or family book
• Photocopy of judicial resolution in the case of guardian or temporary guardian.
2. If the father, mother or legal guardian cannot take the child or adolescent for vaccination, what documents must the person accompanying the child present?
The adult accompanying the child or adolescent may sign a sworn statement certifying parentage, a photocopy of both must be attached.
3. If the child or adolescent does not have an identity card, can they be vaccinated? What documents must they present?
You can access the vaccination with the presentation of the birth certificate or the certificate of live birth and the sworn statement by the responsible adult that accompanies you.
4. Can you get vaccinated if you have nasal congestion or the famous “tĩ syry”?
Yes, you can get vaccinated. Nasal congestion or tĩ syry does not represent a contraindication for vaccination against COVID-19.
5. How long after receiving a vaccine from the regular vaccination schedule can the anti-COVID-19 be applied?
If you receive a dose of any vaccine from the regular schedule, you must wait 2 weeks to apply the dose against COVID-19. Also, if you receive the COVID-19 vaccine, you must wait 2 weeks to receive another vaccine from the regular schedule.
6. How long after applying the yellow fever dose can you receive the COVID-19 vaccine?
If you receive a dose against yellow fever, you must wait 2 weeks to apply the anti-COVID-19.
7. What are the adverse effects that vaccination can cause?
As with any vaccine, there may be associated adverse events that are generally mild and resolve spontaneously. Most common adverse events: pain at the injection site, tiredness, headache, muscle aches, chills, joint pain, fever and swelling at the injection site.
Severe adverse events identified post authorization of the vaccine were infrequent, such as anaphylactic reaction. That is why post-vaccination observation (waiting at the vaccination site) for a period of 30 minutes is recommended, so that, if it occurs, the appropriate treatment can be carried out.
If you have an effect supposedly attributable to vaccination, record that information in http://www.vaccinate.gov.py, in section Do you present any adverse effect to the vaccine? This will help closely monitor the safety of the vaccines.
8. How long after having COVID-19 can the vaccine be given?
The person with COVID-19 should postpone their vaccination for 4 weeks:
– For mild cases or without symptoms, count from the time of diagnosis of the disease
– For cases that required hospitalization, count from the medical discharge.
9. When should I get vaccinated if I have an acute illness?
The person with any acute illness (respiratory symptoms or symptoms with fever) should be vaccinated once they have fully recovered.
10. What are the vaccines that children from 5 to 11 years of age can receive?
Two vaccine platforms are currently being used in several countries, both authorized for emergency use by international and/or national drug regulatory agencies, these are: Pfizer’s mRNA Platform Vaccine and inactivated virus Platform Vaccine: from Sinovac laboratories ( CoronaVac) and Sinopharm.
The Chilean Health Institute (ISP) granted authorization for the emergency use of the CoronaVac vaccine for children aged 6 to 17 years, in September 2021 and, in November, this announcement was extended to the age range for boys and girls from 3 years.
The National Medicines Agency of Argentina (ANMAT) granted authorization for the emergency use of the SINOPHARM vaccine, in October 2021, for children and adolescents aged 3 to 17 years.
In Paraguay, DINAVISA authorized the use of CORONAVAC for children and adolescents from 5 to 17 years of age, in December 2021. The scheme of this platform is two doses, with an interval of 4 weeks.
The United States Regulatory Agency (FDA) granted authorization for emergency use of the Pfizer vaccine for children 5 to 11 years of age, in October 2021. The scheme of this platform is two doses, with an interval of 3 weeks.
11. Is the vaccine safe?
Yes, all vaccines are safe and effective, this claim is based on scientific evidence.
Regarding vaccination in boys and girls under 12 years of age, there is currently data from a phase ½ clinical study, carried out in China, in people aged 3 to 17 years that evaluated safety, tolerability and immunogenicity, and it was determined that CoronaVac it was well tolerated and safe and induced a humoral response in this group, with an excellent antibody response (greater than 90%).
The benefits of Comirnaty (Pfizer) in boys and girls ages 5 to 11 outweigh the risks, especially in those with conditions that increase the risk of severe COVID-19. The clinical trial has been carried out under a double-blind design and of the 1,305 minors who received the vaccine, only three developed the disease, compared to 16 of the 663 who received the placebo, showing an efficacy of 90.7%.